Professional raw steroids suppliers, the company headquarters in xiamen, China. Has a distribution center in Canada, Provide domestic delivery for all canada customers.
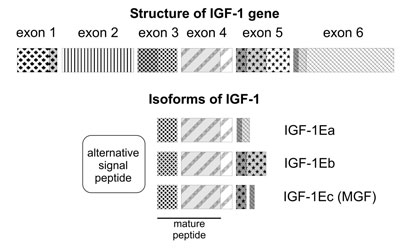
Mechano growth factor (MGF) is a novel splice variant of the Insulin-Like Growth Factor-1 (IGF-1), also known as IGF-1 Ec in humans and IGF-1Eb in rodents. It is actually originally called MGF because the RNA form of it is expressed in muscle tissues in response to the overload or/and damage of muscle growth tissue. The C-terminal peptide of the mechano growth factor (MGF) is a crucial region for the alternative splicing of the peptide. The alternative splicing in the MGF is brought about by the shift in the reading frame in which a specific C-terminal sequence (E-domain) is encoded by exon 5 and the first part of the exon 6. Another interesting point in MGF is that, because of the E domain it contains, MGF can act on muscles independently from the rest of the molecule. Furthermore, MGF can elicit very different effects with mGF promoting satellite cells proliferation and IGF-1 inducing differentiation (Dluzniewska et al. 2005).
Sequence: Tyr-Gln-Pro-Pro-Ser-Thr-Asn-Lys-Asn-Thr-Lys-Ser-Gln-Arg-Arg-Lys-Gly-Ser-Thr-Phe-Glu-Glu-Arg-Lys
Molecular formula: C121H200N42O39
Molar Mass: 2867.14
CAS number: N/A
Synonyms: Mechano Growth Factor, IGF-1 Ec
MGF was suggested to play a number of physiological roles because the failure in its expression may result to age-related loss of skeletal function. Included in its functions is its ability to become a potent neuroprotective as supported by the study that has shown functional copies of the MGF cDNA to be expressed in a plasmid vector which then protected facial neurons after nerve damage (Dluzniewska et al. 2005). One such failure is called sarcopenia. It would also appear that with regards to age, the young have a better ability to respond to MGF (4), and that the elderly experience a decreased response to MGF which results in a decreased ability to stimulate the growth of new muscle tissue.
When mechanical overload is introduced to a muscle (as by weight training), the IGF-1 gene is released and is differentially spliced during the bodies response. Initially, it it is spliced to produce predominantly IGF-1Ec (called the MGF splice variant of IGF-1). This early splicing stimulates satellite cells into activation. Which in turn allows the activation of extra undamaged nuclei to grow new muscle fiber and tissue. The appearance of MGF also initiates the upregulation of new protein synthesis. After this initial splicing of IGF-1 into MGF, production then switches towards producing a systemic release of IGF-1Ea from the liver, which also upregulates protein synthesis as well. The expression of IGF-1 splice variants, over the course of the healing and regrowth phase of muscle repair is thought to be the primary mechanism by which the body produces new muscle tissue.
Insulin like growth factor-1 (IGF-1) expression is
implicated in myocardial pathophysiology, and two IGF-1 mRNA splice
variants have been detected in rodents, IGF-1Ea and mechano-growth
factor (MGF). Recent research has shown that the expression pattern of
IGF-1 gene transcripts in rat myocardium from 1 h up to 8 wks after
myocardial infarction induced by left anterior descending coronary
artery ligation. In addition, we characterized IGF-1 and MGF E peptide
action and their respective signaling in H9C2 myocardial-like cells in
vitro. IGF-1Ea and MGF expression were significantly increased, both at
transcriptional and translational levels, during the late postinfarction
period (4 and 8 wks) in infarcted rat myocardium. Measurements of serum
IGF-1 levels in infarcted rats were initially decreased (24 h up to 1
wk) but remained unaltered throughout the late experimental phase (4 to 8
wks) compared with sham-operated rats. Furthermore, specific
anti–IGF-1R neutralizing antibody failed to block the synthetic MGF E
peptide action, whereas it completely blocked IGF-1 action on the
proliferation of H9C2 cells. Moreover, this synthetic MGF E peptide did
not activate Akt phosphorylation, whereas it activated ERK1/2 in H9C2
rat myocardial cells. These data support the role of IGF-1 expression in
the myocardial repair process and suggest that synthetic MGF E peptide
actions may be mediated via an IGF-1R independent pathway in rat
myocardial cells, as suggested by in vitro experiments.
Recent
studies have also discovered two clones of the hybridonoma secreting
monoclonal antibodies to the mechano-growth factor have been developed
by cell fusion technique. The monoclonal antibody of one clone
recognizes the human MGF peptide that is absent in insulin-like growth
factor-1 (IGF-1) which comprised mostly of amino acids from 87-111.
Enzyme-linked immunosorbent assay (ELISA) has further shown that there
is a high affinity binding constants with the full length of the MGF and
the 87-111 fragments of the clones. These can then be used for the
quantitation of the MGF through sandwich type assay (Kravchenko et al.
2006).
